 Not require to lose or gain electrons to complete its energy shell i.e. Noble gases like neon have two valence electrons so it does Possess both positive and negative valency, and atoms having eight outermostĮlectrons, valency will be zero (i.e. Outermost shells is between one to four, the atom has positive valency and ifĮlectrons are between four to eight, the valency is calculated by subtractingįrom eight and valency will be zero. Lost, gained, or shared with another atom at the time of bond formation.Īn atom is said to be stable when its outermost shells haveĮight electrons (except H & He). The valency of an atom is determined based on the number of electrons Valenceĭescribes how easily an atom or a free radical can combine with other chemical There are many different ways to find out the valency of anĪtom which reflects the ability of an atom to bond with other atoms. Of an atom is called valence electrons, and there is only one electron present  
The total number of electrons present in the valence shell Of n, and in 2s☢p⁶, the highest value of n is 2 so that the valence shell of Ne The highest number of principle quantum numbers which is expressed in the term Neon electron configuration Ne (10) = 1s 2 2s☢p⁶ (completeĪs we know, the valence shell of an atom can be found from Again another two electrons will go in 2s orbital and the remaining 6 will go to the 2p orbital. the first two electrons will go in the 1s orbital as the S orbital can hold a maximum of 2 electrons only. The neon atom has a total of 10 electrons so, we have to put 10 electrons in orbitals. Less) in any condition for a particular atom and may or may not be equal to itsĮlectron configuration is the arrangement of electrons on The valenceĮlectron for a neutral atom is always definite, it cannot be varied (more or Valence electrons are the total number of electrons present In order to achieve this full octet, potassium transfers its one valence electron causing bromine to have 8 valence electrons or, a full octet/closed shell. You? But before that let's have some ideas about what these terms are: Difference Between Valence Electrons and Valency For Cl, Z 17 and the electron configuration is 1 s2 2 s2 2 p6 3 s2 3 p5. For example, consider a 2 s electron of Cl. You are here to know valence electrons and valency of neon, aren’t Note that while we often refer to the Z eff of a valence electron, we can calculate the Z eff for any electron by taking into account only the number of core electrons that are shielding. Indicators, fluorescent lamps, and as a cryogenic in refrigerators. Neon-20, neon-22, and neon-21Īre the most stable isotopes of neon. 
Asįor now, It has more than 12 isotopes discovered. Till now, not any stable compounds of neon is discovered. It is widely used in fluorescent lamps and electric The second lightest noble gas after hydrogen and glows reddish-orange on the Is colorless, odorless, tasteless, and lighter than air inert gas. A fun video showing potassium metal and its reactivity can be found HERE.Neon, a chemical element with the symbol He and atomic number 10, It is used to create this same effect and color in fireworks. Potassium burns with a purple flame and throws purple sparks. (Make sure you visit this, because I have mentioned the valence electrons along with the images for each element). But if you want to see the valence electrons of all the 118 elements, then visit: Valence electrons of all the elements. The heat of this reaction ignites the hydrogen gas that is also produced from the water. These are the Valence electrons of first 20 Elements. Pure potassium metal reacts explosively when placed in water as it donates its valence electron to oxygen. (Only the 3 outer shells are depicted here.) Neutral potassium (K) atom (L) compared to the much smaller potassium (K +) ion (R). That is why potassium forms a 1+ ionic state so violently. With a lower ionization energy, potassium will give up its valence electron more eagerly than sodium in an ionic interaction, in order to reach the stability of the 3 s 23 p 6 noble gas configuration of argon, which is a multi-di-electron state with three concentric full shells. These orbitals represent phase-locked, resonant, coherent, harmonic, stationary waves. 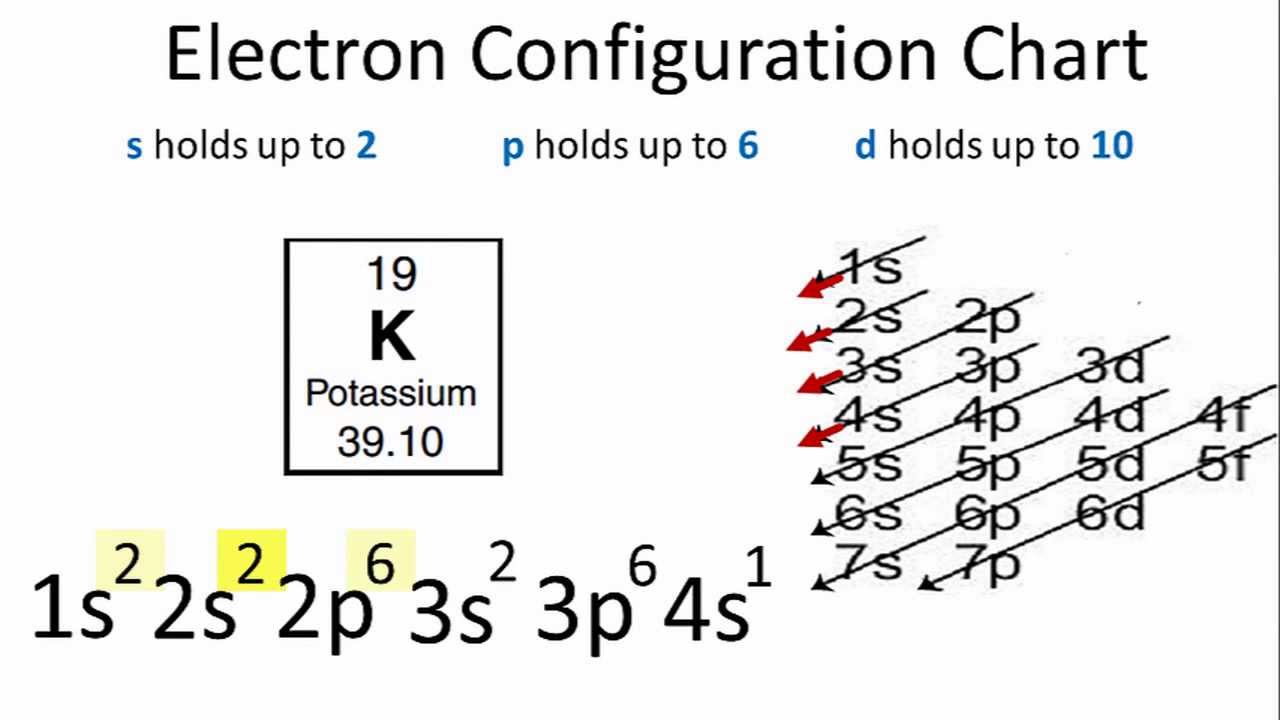
CLICK HERE to interact with this object.Īs we saw in the case of argon, the 3 rd shell orbitals are more like spherical tetrahedra, and the 4 th shell is a single electron in a spherical s-orbital. Being one shell larger than sodium, potassium has a lower ionization energy and is therefore more reactive. Potassium has the same electron configuration as sodium, but with three full shells within that have the identical configuration to argon. Potassium atoms have 19 electrons and the electronic shell structure is 2, 8, 8, 1 with Atomic Term Symbol (Quantum Numbers) 2S1/2. It has 19 protons and 20 neutrons in the nucleus, giving it a mass of 39 amu, and it has 19 electrons enveloping the nucleus. Potassium is the 19 th element on the periodic table. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
